The Lowdown
Notoriously difficult to diagnose, neutropenic sepsis kills 3 people every day in the UK. It’s a whole-body reaction to an infection, which can quickly become life-threatening. Considered a medical emergency, it often affects cancer patients as chemotherapy can cause a low neutrophil (white blood cell) count.
People with neutropenic sepsis need urgent diagnosis and treatment with antibiotics. But currently, only 33% of diagnosed patients get medical treatment within an hour. And this significantly increases its mortality rate.
Apart from the devastating loss of life, the low diagnosis and treatment rates cost global health systems a phenomenal amount of money. A recent study found sepsis complications can double overall cancer care costs.
52North zeroed in on a clear and urgent need to develop a more effective pathway to faster diagnosis.

The Problem
Recognising that neutropenic sepsis (NS) is the most fatal side-effect of chemotherapy treatment, 52North wanted to develop a test to help these people. The advice to cancer patients is to go straight to A&E if they feel unwell. But there are problems with this:
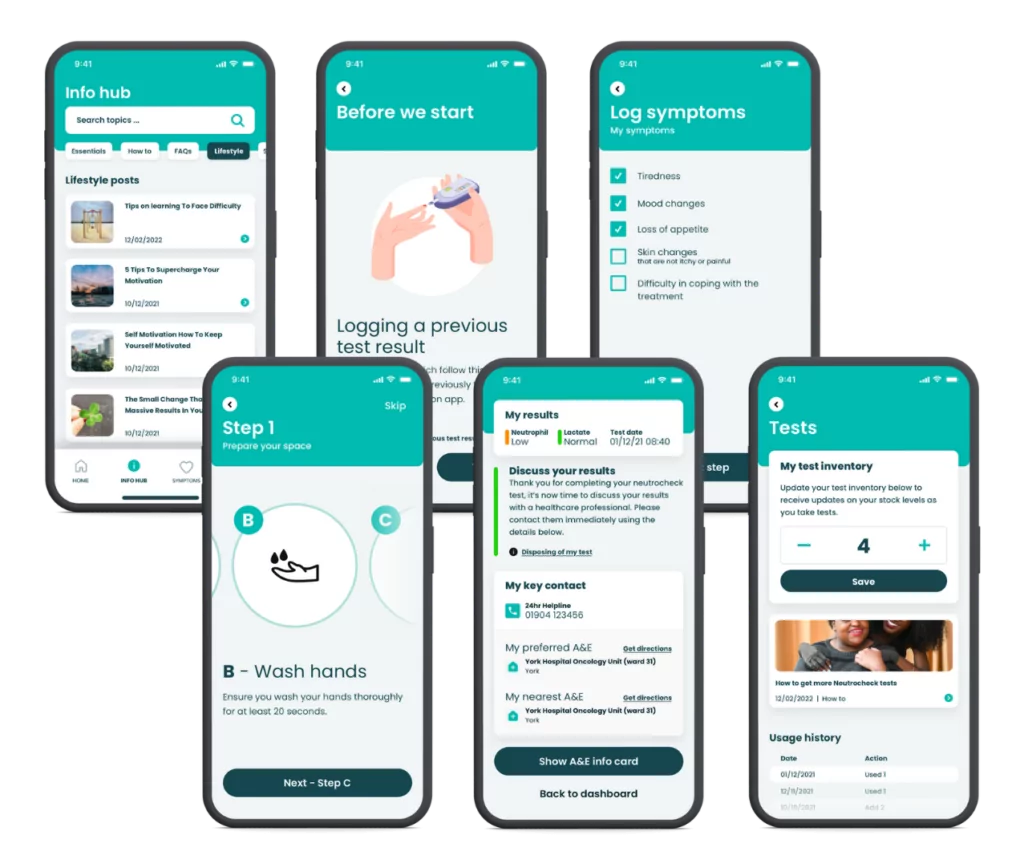
54North developed Neutrocheck®, a low-cost finger prick blood test to give a rapid and reliable result. They approached us to help develop a companion app to increase successful testing procedures, record results, and offer additional support.
The Solution
We worked closely with 52North and Macmillan Cancer Support to design an app to accompany their rapid testing kits.
Our strategy for success focused on the need to increase the adoption and success rate of sepsis diagnosis, offering additional support to cancer patients.
Requirements
- Patient engagement
- We needed to optimise patient onboarding and engagement with the tests.
- Test-to-app ratio
- We wanted to maximise the ratio between tests issued and results recorded on the app.
- Increased test reliability
- 52North was also keen that we optimise the test results to make sure the app process offers users a notable benefit.
- Accessibility of data
- Another key objective was to make sure we could maximise user data completion. Profile completion needed to be based on relevant user data in a consistent format.
The Product
- Discovery
- During this first phase, we were highly engaged with user groups and Macmillan to walk through the practicalities.
- Design
- This next phase involved a lot of collaboration with 52North and Macmillan to check that the designs would work and support the testing experience for patients.
- Build
- Once all designs were approved, we moved to build a minimum viable product (MVP) to test the market before full rollout.